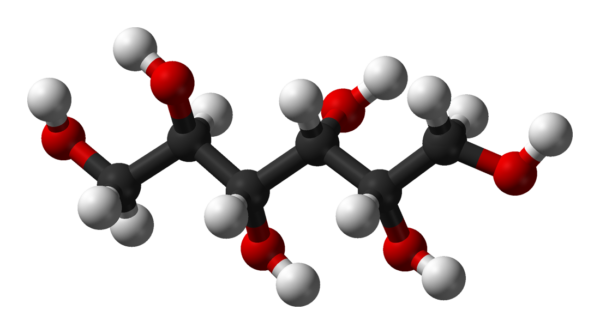
Sorbitol
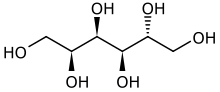
Sorbitol less commonly known as glucitol , is a sugar alcohol with a sweet taste which the human body metabolizes slowly. It can be obtained by reduction of glucose, which changes the converted aldehyde group (−CHO) to a primary alcohol group (−CH2OH). Most sorbitol is made from potato starch, but it is also found in nature, for example in apples, pears, peaches, and prunes.[4] It is converted to fructose by sorbitol-6-phosphate 2-dehydrogenase. Sorbitol is an isomer of mannitol, another sugar alcohol; the two differ only in the orientation of the hydroxyl group on carbon 2. While similar, the two sugar alcohols have very different sources in nature, melting points, and uses.
As an over-the-counter drug, sorbitol is used as a laxative to treat constipation
Application
It is used as a a sweetening agent, food additive, toothpaste, tobacco, toiletries and in cosmetics. It is used for vitamin-C fermentation. It is used as a excipient and intravenous osmotic diuretic in pharmaceutical fields. It is also used in the manufacture of polyethers for polyurethanes and surfactants.
| Chemical formula | C6H14O6 |
|---|---|
| Molar mass | 182.17 g/mol |
| Appearance | White crystalline powder |
| Density | 1.49 g/cm3 |
| Melting point | 94–96 °C (201–205 °F; 367–369 K)[2] |
| Solubility in water | 2350 g/L |
| log P | -4.67 |
| Magnetic susceptibility (χ) | -107.80·10−6 cm3/mol |


Reviews
There are no reviews yet.