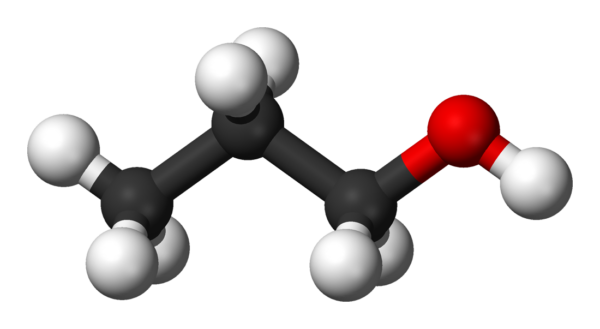
N-Propanol
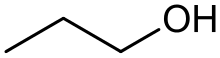
Propan-1-ol (also propanol, n-propyl alcohol) is a primary alcohol with the formula CH3CH2CH2OH and sometimes represented as PrOH or n-PrOH. It is a colourless liquid and an isomer of 2-propanol. It is formed naturally in small amounts during many fermentation processes and used as a solvent in the pharmaceutical industry, mainly for resins and cellulose esters, and, sometimes, as a disinfecting agent.
Propan-1-ol is manufactured by catalytic hydrogenation of propionaldehyde. Propionaldehyde is produced via the oxo process by hydroformylation of ethylene using carbon monoxide and hydrogen in the presence of a catalyst such as cobalt octa carbonyl or a rhodium complex
Application
Normal propanol is used to manufacture other solvents such as cosmetics, skin/hair preparations, perfumes, pharmaceuticals, lacquer formulations, dye solutions, antifreezes, soaps and window cleaner. It can also be used to create printing inks, coatings, de-greasing fluids, adhesives, insecticides and herbicides. Normal propanol is also used as a chemical intermediate to create esters, halides, propyl amines and propyl acetate. The end user markets of this product are the cosmetics, cleaning, motor, printing, coatings and chemical industries.
| Chemical formula | C3H8O |
|---|---|
| Molar mass | 60.096 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | mild, alcohol-like |
| Density | 0.803 g/mL |
| Melting point | −126 °C; −195 °F; 147 K |
| Boiling point | 97 to 98 °C; 206 to 208 °F; 370 to 371 K |
| Solubility in water | miscible |
| log P | 0.329 |
| Vapor pressure | 1.99 kPa (at 20 °C) |
| Acidity (pKa) | 16 |
| Basicity (pKb) | −2 |
| Magnetic susceptibility (χ) | −45.176·10−6 cm3/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 1.959 mPa·s (at 25 °C) |
| Dipole moment | 1.68 D |


Reviews
There are no reviews yet.