
Mixed Xylene
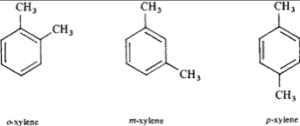
In organic chemistry, xylene or xylol (from Greek ξύλον (xylon) ‘wood’; IUPAC name: dimethylbenzene) are any of three organic compounds with the formula (CH3)2C6H4. They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are substituted determines which of three structural isomers results. It is a colorless, flammable, slightly greasy liquid of great industrial value.
The mixture is referred to as both xylene and, more precisely, xylenes. Mixed xylenes refers to a mixture of the xylenes plus ethylbenzene. The four compounds have identical empirical formulas C8H10. Typically the four compounds are produced together by various catalytic reforming and pyrolysis methods.
Application
Ortho Xylene is used as solvent in printing, rubber, and leather industries. In thinning paints and varnishes, it can be substituted for toluene where slower drying is desired. It is used in the production of plastic bottles and polyester clothing. Used as solvent in printing, rubber, and leather industries. In thinning paints and varnishes, it can be substituted for toluene where slower drying is desired.
| Molecular formula | C8H10 |
|---|---|
| SMILES | Cc1c(C)cccc1 |
| Molar mass | 106.16 g/mol |
| Appearance | Clear, colorless liquid |
| CAS number | [1330-20-7] |
| Density and phase | 0.864 g/mL, liquid |
| Solubility in water | Practically insoluble |
| Melting point | −47.4 °C (−53.3 °F; 226 K) |
| Boiling point | 138.5 °C (281.3 °F; 412 K) |
| Viscosity | 0.812 cP at 20 °C (68 °F) |



Reviews
There are no reviews yet.