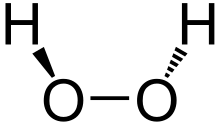
Hydrogen Per Oxide
Hydrogen peroxide is a chemical compound with the formula H2O2. In its pure form, it is a very pale blue[5] liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or “high-test peroxide”, decomposes explosively when heated and has been used both as a monopropellant and an oxidizer in rocketry.[6]
Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly into water and elemental oxygen when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a stabilizer in a weakly acidic solution in an opaque bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases.
Application
Used in water treatment industry, pulp and paper industry and as antimicrobial agent.
| Chemical formula | H2O2 |
|---|---|
| Molar mass | 34.014 g·mol−1 |
| Appearance | Very light blue liquid |
| Odor | slightly sharp |
| Density | 1.11 g/cm3 (20 °C, 30% (w/w) solution)[1] |
| Melting point | −0.43 °C (31.23 °F; 272.72 K) |
| Boiling point | 150.2 °C (302.4 °F; 423.3 K) (decomposes) |
| Solubility in water | Miscible |
| Solubility | soluble in ether, alcohol |
| log P | -0.43[2] |
| Vapor pressure | 5 mmHg (30 °C)[3] |
| Acidity (pKa) | 11.75 |
| Magnetic susceptibility (χ) | −17.7·10−6 cm3/mol |
| Refractive index (nD) | 1.4061 |
| Viscosity | 1.245 cP (20 °C) |
| Dipole moment | 2.26 D |




Reviews
There are no reviews yet.