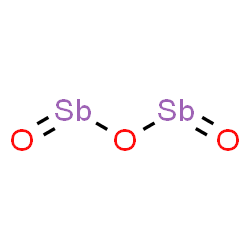
Antimony Tri Oxide

Antimony(III) oxide is the inorganic compound with the formula Sb2O3. It is the most important commercial compound of antimony. It is found in nature as the minerals valentinite and senarmontite. Like most polymeric oxides, Sb2O3 dissolves in aqueous solutions with hydrolysis. A mixed arsenic-antimony oxide occurs in nature as the very rare mineral stibioclaudetite.
Application
Flame retardant for wide range of plastics, rubbers, paper and textiles . Catalyst in PET production. Activator in glass industry. Flocculant in Titanium Dioxide production. Paints and Adhesives industries. Pigments, Ceramic frites
| Chemical formula | Sb2O3 |
|---|---|
| Molar mass | 291.518 g/mol |
| Appearance | white solid |
| Odor | odorless |
| Density | 5.2 g/cm3, α-form |
| Melting point | 656 °C (1,213 °F; 929 K) |
| Boiling point | 1,425 °C (2,597 °F; 1,698 K) (sublimes) |
| Solubility in water | 370±37 µg/L between 20.8 °C and 22.9 °C |
| Solubility | soluble in acid |
| Magnetic susceptibility (χ) | −69.4×10−6 cm3/mol |
| Refractive index (nD) | 2.087, α-form |


Reviews
There are no reviews yet.